


UNLOCKING PHENOTYPIC PLASTICITY
Cancer is a moving target causing:
- Resistance mechanism
- Immune escape
- Relapsing process

Adapted from Hallmarks of cancer: New dimension*


CURRENT TREATMENT MUST
BE COMBINED TO BE EFFICIENT
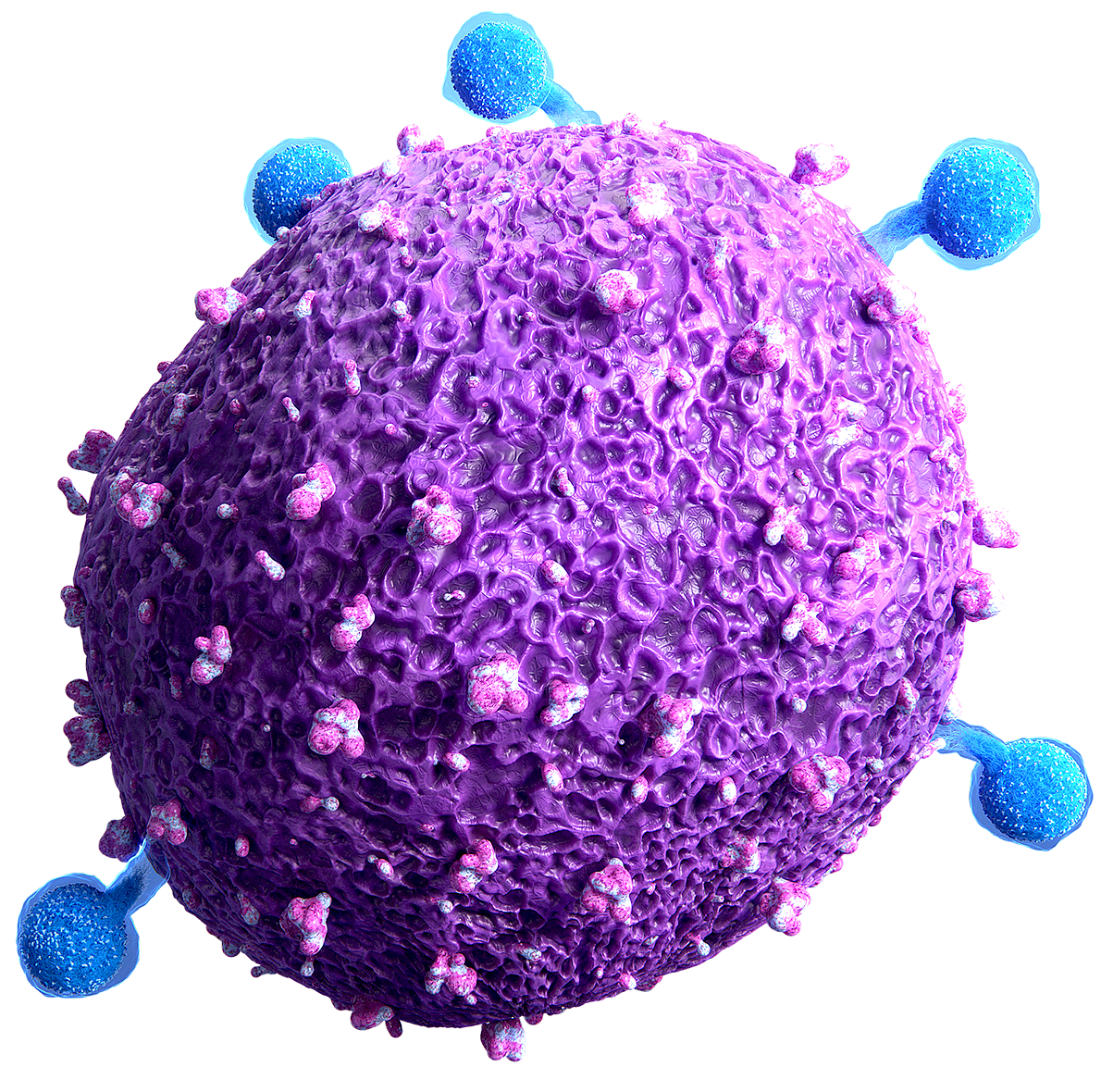
LACK OF ANTIGENICITY
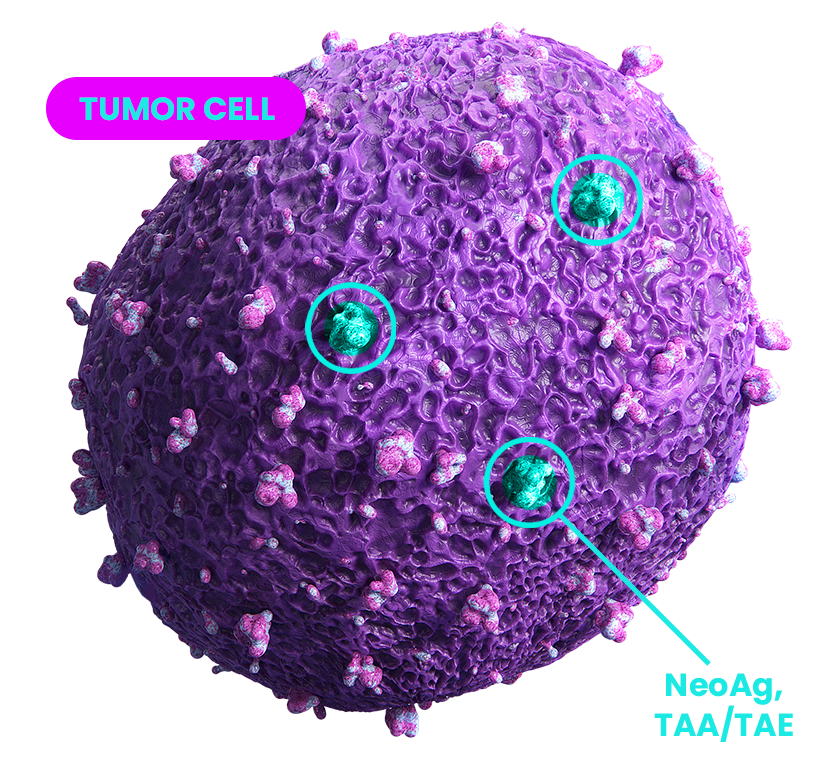
LACK OF ANTIGENICITY
BRENUS
PHARMA:
THE MISSING LINK ?
Brenus Pharma develops a next gen immun therapeutic based on adaptive & multispecific targets to educate the immune system to anticipate tumor plasticity & treatment resistance.
ENGAGE WITH US IN
A CHANGE OF PARADIGM
EFFICIENT TECHNOLOGY
Anticipates risks of tumor escape by targeting over 250 cancer-related antigens driven by relapse mechanism.
Answers huge unmet need in solid tumors : Platform strategy, combining impactful technologies, with standardized steps for a quicker generation of immunotherapy for solid tumors
DERISKED APPROACH
2nd generation of innovative cancer vaccine strategy : previous haptenized approaches show significative historical clinical DATA (Glioblastoma + Melanoma).
Preclinical package endoresed with FDA Pre-IND passed.
GMP manufacturing ready.
Top international scientific leader on board & experienced governance in Healthtech industries.
LIMITLESS POTENTIAL
IP by design-platform strategy ; 22 patent granted.
Tailored first-in-class drugs design.
Dynamic immuno-oncology market with mRNA and Personalized Cancer Vaccine.
Ensuring treatment’s availability to a wide population of patient in therapeutic issue.





